We work in neuroscience, immunology, metabolic disease, and more.
TwinRCTsTM
Clinical trials powered by patients’ digital twins
Shorten time to enrollment in late stage studies
Shorten time to enrollment in late stage studies
TwinRCTs reach full enrollment sooner because they require fewer patients to achieve the same power as traditional trial designs.

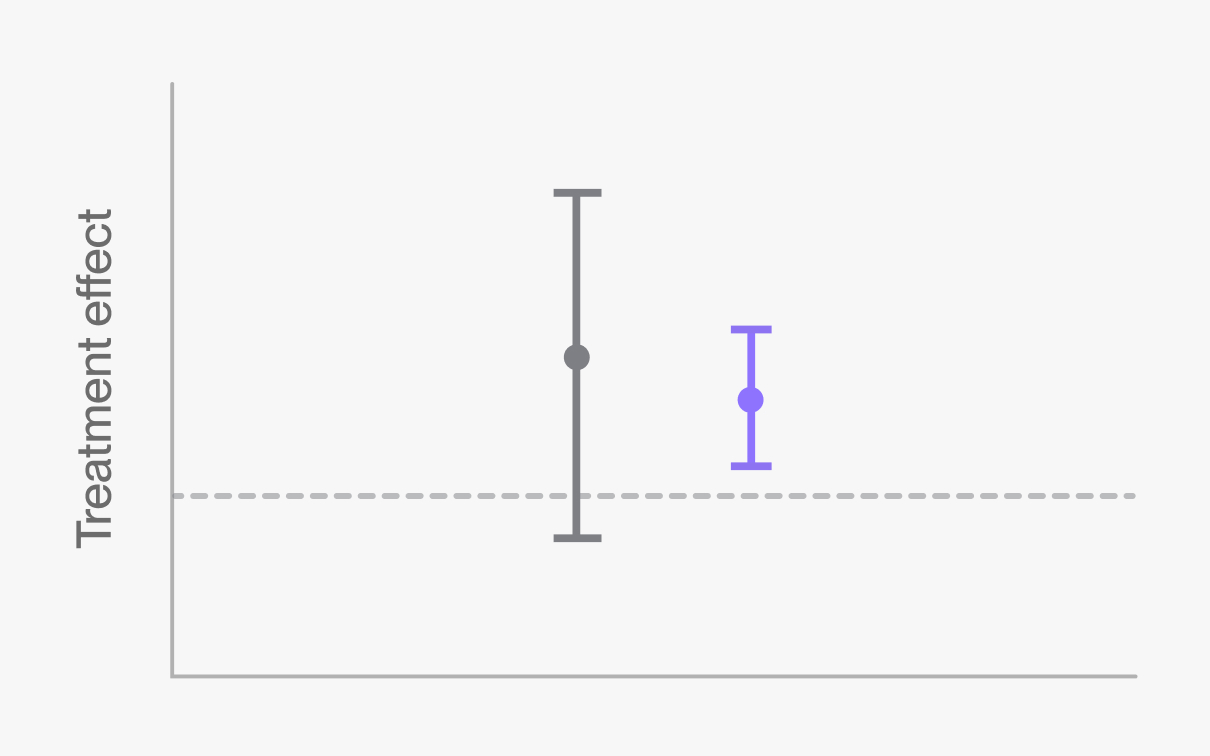
Make confident decisions from early stage studies
Make confident decisions from early stage studies
TwinRCTs improve the ability to observe treatment effects in early stage studies by increasing power without adding more patients.
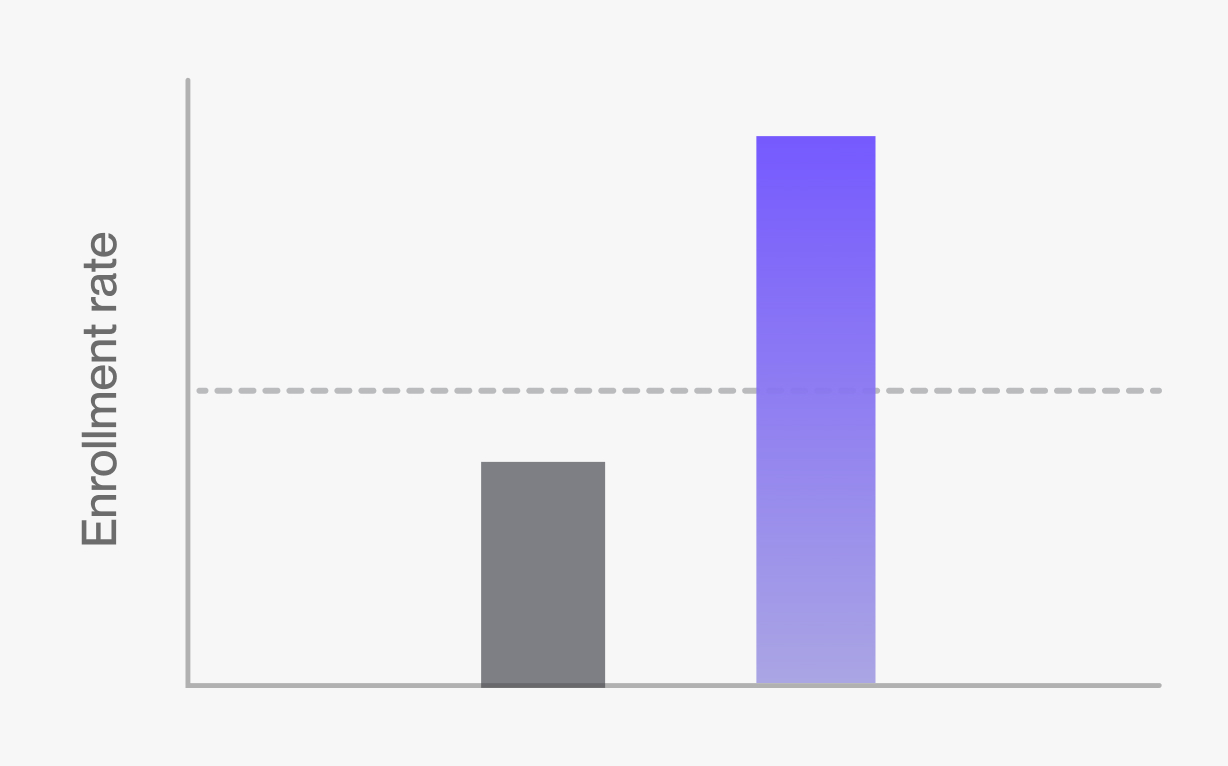
Attract study participants
Attract study participants
TwinRCTs give participants a greater chance of receiving the experimental treatment because they use smaller control groups.
We're solving AI for medicine
What are digital twins?

What are digital twins?
A patient’s digital twin is a comprehensive forecast of their future health. We invent and deploy new types of generative models trained on extensive patient-level data from previous studies.
A participant's baseline data is collected.
Their baseline data is run through our AI model trained on historical data.
A participant's digital twin is created.
How do digital twins enhance clinical trials?

How do digital twins enhance clinical trials?
We calculate prognostic scores for each patient in a randomized clinical trial using their digital twins. Adjusting for these scores in the analysis increases power while adhering to guidance from the US Food and Drug Administration and European Medicines Agency.
At the start of a trial, each participant has one of two potential outcomes: one outcome if they are assigned to the treatment group or one if they are assigned to the control group.
Their digital twin forecasts their potential control group outcome regardless of their actual assignment in the trial.
Participants’ digital twins are used in TwinRCTs, highly powered trials with smaller control groups.



